Abstract
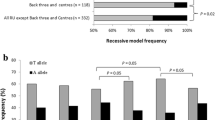
Peroxisome proliferator-activated receptor α (PPARα) regulates genes responsible for skeletal and heart muscle fatty acid oxidation. Previous studies have shown that the PPARα intron 7 G/C polymorphism was associated with left ventricular growth in response to exercise. We speculated that GG homozygotes should be more prevalent within a group of endurance-oriented athletes, have normal fatty acid metabolism, and increased percentages of slow-twitch fibers. We have tested this hypothesis in the study of a mixed cohort of 786 Russian athletes in 13 different sporting disciplines prospectively stratified by performance (endurance-oriented athletes, power-oriented athletes and athletes with mixed endurance/power activity). PPARα intron 7 genotype and allele frequencies were compared to 1,242 controls. We found an increasing linear trend of C allele with increasing anaerobic component of physical performance (P=0.029). GG genotype frequencies in endurance-oriented and power-oriented athletes were 80.3 and 50.6%, respectively, and were significantly (P<0.0001) different compared to controls (70.0%). To examine the association between PPARα gene variant and fiber type composition, muscle biopsies from m. vastus lateralis were obtained and analyzed in 40 young men. GG homozygotes (n=25) had significantly (P=0.003) higher percentages of slow-twitch fibers (55.5±2.0 vs 38.5±2.3%) than CC homozygotes (n=4). In conclusion, PPARα intron 7 G/C polymorphism was associated with physical performance in Russian athletes, and this may be explained, in part, by the association between PPARα genotype and muscle fiber type composition.


Similar content being viewed by others
References
Allard MF, Schonekess BO, Henning SL, English DR, Lopaschuk GD (1994) Contribution of oxidative metabolism and glycolysis to ATP production in hypertrophied hearts. Am J Physiol Heart Circ Physiol 267:742–750
Aoyama T, Peters JM, Iritani N, et al. (1998) Altered constitutive expression of fatty acid-metabolizing enzymes in mice lacking the peroxisome proliferator-activated receptor α (PPARα). J Biol Chem 273:5678–5684
Barger PM, Brandt JM, Leone TC, Weinheimer CJ, Kelly DP (2000) Deactivation of peroxisome proliferator-activated receptor α during cardiac hypertrophic frowth. J Clin Invest 105:1723–1730
Bolla MK, Haddad L, Humphries SE, Winder AF, Day INM (1995) A method of determination of hundreds of APOE genotypes utilizing highly simplified, optimized protocols and restriction digestion analysis by microtitre array diagonal gel electrophoresis (MADGE). Clin Chem 41:1599–1604
Braissant O, Foufelle F, Scotto C, Dauca M, Wahli W (1996) Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, -beta, and -gamma in the adult rat. Endocrinology 137:354–366
Brooks GA, Mercier J (1994) Balance of carbohydrate and lipid utilization during exercise: the crossover concept. J Appl Physiol 76:2253–2261
Cresci S, Wright LD, Spratt JA, Briggs FN, Kelly DP (1996) Activation of a novel metabolic gene regulatory pathway by chronic stimulation of skeletal muscle. Am J Physiol Cell Physiol 270:1413–1420
Flavell DM, Ireland H, Stephens JW et al (2005) Peroxisome proliferator-activated receptor α gene variation influences age of onset and progression of type 2 diabetes. Diabetes 54:582–586
Flavell DM, Jamshidi Y, Hawe E et al (2002) Peroxisome proliferator-activated receptor α gene variants influence progression of coronary atherosclerosis and risk of coronary artery disease. Circulation 105:1440–1445
Flavell DM, Pineda Torra I, Jamshidi Y et al (2000) Variation in the PPARα gene is associated with altered function in vitro and plasma lipid concentrations in type II diabetic subjects. Diabetologia 43:673–680
Foucher C, Rattier S, Flavell DM, Talmud PJ, Humphries SE, Kastelein JJ et al (2004) Response to micronized fenofibrate treatment is associated with the peroxisome-proliferator-activated receptors alpha G/C intron 7 polymorphism in subjects with type 2 diabetes. Pharmacogenetics 14(12):823–829
Gulick T, Cresci S, Caira T, Moore DD, Kelly DP (1994) The peroxisome proliferator-activated receptor regulates mitochondrial fatty acid oxidative enzyme gene expression. Proc Natl Acad Sci USA 91:11012–11016
Horowitz JF, Leone TC, Feng W, Kelly DP, Klein S (2000) Effect of endurance training on lipid metabolism in women: a potentional role for PPARα in the metabolic response to training. Am J Physiol Endocrinol Metab 279:348–355
Jamshidi Y, Montgomery HE, Hense H-W et al (2002) Peroxisome proliferator-activated receptor α gene regulates left ventricular growth in response to exercise and hypertension. Circulation 105:950–955
Kagaya Y, Kanno Y, Takeyama D et al (1990) Effects of long-term pressure overload on regional myocardial glucose and free fatty acid uptake in rats. A quantitative autoradiographic study. Circulation 81:1353–1361
Russel AP, Feilchenfeldt Y, Schreiber S et al (2003) Endurance training in humans leads to fiber type-specific increases in levels of peroxisome proliferator-activated receptor-γ coactivator-1 and peroxisome proliferator-activated receptor-α in skeletal muscle. Diabetes 52:2874–2881
Schmitt B, Fluck M, Decombaz J (2003) Transcriptional adaptations of lipid metabolism in tibialis anterior muscle of endurance-trained athletes. Physiol Genom 15:148–157
Vohl M-C, Lepage P, Gaudet D et al (2000) Molecular scanning of the human PPARα gene: association of the L162V mutation with hyperapobetalipoproteinemia. J Lipid Res 41:945–952
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmetov, I., Mozhayskaya, I.A., Flavell, D.M. et al. PPARα gene variation and physical performance in Russian athletes. Eur J Appl Physiol 97, 103–108 (2006). https://doi.org/10.1007/s00421-006-0154-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-006-0154-4