Abstract
Members of the cytosolic sulfotransferase (SULT) superfamily catalyse the sulfation of a multitude of xenobiotics, hormones and neurotransmitters. Humans have at least 10 functional SULT genes, and a number of recent advances reviewed here have furthered our understanding of SULT function. Analysis of expression patterns has shown that sulfotransferases are highly expressed in the fetus, and SULTs may in fact be a major detoxification enzyme system in the developing human. The X-ray crystal structures of three SULTs have been solved and combined with mutagenesis experiments and molecular modelling, they have provided the first clues as to the factors that govern the unique substrate specificities of some of these enzymes. In the future these and other studies will facilitate prediction of the fate of chemicals metabolised by sulfation. Variation in sulfation capacity may be important in determining an individual's response to xenobiotics, and there has been an explosion in information on sulfotransferase polymorphisms and their functional consequences, including the influence of SULT1A1 genotype on susceptibility to colorectal and breast cancer. Finally, the first gene knockout experiments with SULTs have recently been described, with the generation of estrogen sulfotransferase deficient mice in which reproductive capacity is compromised. Our improved understanding of these enzymes will have significant benefits in such diverse areas as drug design and development, cancer susceptibility, reproduction and development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Notes
Raftogianis RB et al, submitted for publication. In particular, the new nomenclature for the SULT1C enzymes may cause some confusion. The human SULT1C2 referred to here was called SULT1C1 or SULT1C sulfotransferase 1 in the original descriptions,18,19,75 and the human SULT1C4 referred to here was originally called SULT1C2 or SULT1C sulfotransferase.19,75
This may not be the case for liver, where the presence of other SULT isoforms such as 1B1 may interfere with measurement of enzyme activity with non-selective substrates such as 4-nitrophenol (our unpublished work).
Abbreviations
- SULT:
-
Sulfotransferase
- PAPS:
-
3′-phosphoadenosine 5′-phosphosulfate
References
Robbins PW, Lipmann F . Isolation and identification of active sulfate J Biol Chem 1957 229: 837–851
Klaassen CD, Boles JW . The importance of 3′-phosphoadenosine 5′-phosphosulfate (PAPS) in the regulation of sulfation FASEB J 1997 11: 404–418
Dousa MK, Tyce GM . Free and conjugated plasma catecholamines, DOPA and 3-O-methyldopa in humans and various animal species Proc Soc Exp Biol Med 1988 188: 427–434
Eisenhofer G, Coughtrie MWH, Goldstein DS . Dopamine sulphate: an enigma resolved Clin Exp Pharmacol Physiol 1999 26: S41-S53
Ogura K, Kajita J, Narihata H, Watabe T, Ozawa S, Nagata K et al . Cloning and sequence analysis of a rat liver cDNA encoding hydroxysteroid sulfotranferase Bio chem Biophys Res Commun 1989 165: 168–174
Ogura K, Kajita J, Narihata H, Watabe T, Ozawa S, Nagata K et al . cDNA cloning of the hydroxysteroid sulfotransferase STa sharing a strong homology in amino acid sequence with the senescence mark er protein SMP-2 in rat livers Biochem Biophys Res Commun 1990 166: 1494–1500
Watabe T, Ogura K, Satsukawa M, Okuda H, Hiratsuka A . Molecular cloning and functions of rat liver hydroxysteroid sulfotransferases catalysing covalent binding of carcinogenic polycyclic arylmethanols to DNA Chem Biol Interact 1994 92: 87–105
Otterness DM, Wieben ED, Wood TC, Watson RWG, Madden BJ, McCormick DJ et al . Human liver dehydroepiandrosterone sulfotransferase: molecular cloning and expression of cDNA Mol Pharmacol 1992 41: 865–872
Barker EV, Hume R, Hallas A, Coughtrie MWH . Dehydroepiandrosterone sulfotransferase in the developing human fetus—quantitative biochemical and immunological characterization of the hepatic, renal, and adrenal enzymes Endocrinology 1994 134: 982–989
Hume R, Barker EV, Coughtrie MWH . Differential expression and immunohistochemical localization of the phenol and hydroxysteroid sulfotransferase enzyme families in the developing lung Histochem Cell Biol 1996 105: 147–152
Hume R, Coughtrie MWH . Phenolsulfotransferase—localization in kidney during human embryonic and fetal development Histochem J 1994 26: 850–855
Richard K, Hume R, Kaptein E, Stanley EL, Visser TJ, Coughtrie MWH . Sulfation of thyroid hormone and dopamine during human development—ontogeny of phenol sulfotransferases and arylsulfatase in liver, lung and brain J Clin Endocrinol Metab 2001 86: 2734–2742
Stanley EL . Expression and function of iodothyronine metabolising enzymes during human placental and fetal development PhD Thesis University of Dundee 2001
Hakkola J, Pelkonen O, Pasanen M, Raunio H . Xenobiotic-metabolizing cytochrome P450 enzymes in the human feto-placental unit: role in intrauterine toxicity Crit Rev Toxicol 1998 28: 35–72
Coughtrie MWH, Burchell B, Leakey JEA, Hume R . The inadequacy of perinatal glucuronidation—immunoblot analysis of the developmental expression of individual UDP-glucuronosyltransferase isoenzymes in rat and human-liver microsomes Mol Pharmacol 1988 34: 729–735
Varin L, Marsolais F, Richard M, Rouleau M . Biochemistry and molecular biology of plant sulfotransferases FASEB J 1997 11: 517–525
Weinshilboum RM, Otterness DM, Aksoy IA, Wood TC, Her C, Raftogianis RB . Sulfotransferase molecular biology: cDNAs and genes FASEB J 1997 11: 3–14
Her C, Kaur GP, Athwal RS, Weinshilboum RM . Human sulfotransferase SULT1C1: cDNA cloning, tissue-specific expression and chromosomal localization Genomics 1997 41: 467–470
Sakakibara Y, Yanagishita M, Katafuchi J, Ringer DP, Takami Y, Nakayama T et al . Molecular cloning, expression, and characterization of novel human SULT1C sulfotransferases that catalyze sulfonation of N-hydroxy-2-acetylaminofluorene J Biol Chem 1998 273: 33929–33935
Parker CR, Falany CN, Stockard CR, Stankovic AK, Grizzle WE . Immunohistochemical localization of dehydroepiandrosterone sulfotransferase in human fetal tissues J Clin Endocrinol Metab 1994 78: 234–236
Song WC, Melner MH . Editorial: steroid transformation enzymes as critical regulators of steroid action in vivo Endocrinology 2000 141: 1587–1589
Buirchell BJ, Hähnel R . Metabolism of estradiol-17b in human endometrium during the menstrual cycle J Steroid Biochem 1975 6: 1489–1494
Rubin GL, Harrold AJ, Mills JA, Falany CN, Coughtrie MWH . Regulation of sulfotransferase expression in the endometrium during the menstrual cycle, by oral contraceptives and during early pregnancy Mol Human Reprod 1999 5: 995–1002
Tseng L, Liu HC . Stimulation of arylsulfotransferase activity by progestins in human endometrium in vitro J Clin Endocrinol Metab 1981 53: 418–421
Falany JL, Falany CN . Regulation of estrogen sulfotransferase in human endometrial adenocarcinoma cells by progesterone Endocrinology 1996 137: 1395–1401
Kester MHA, Bulduk S, Tibboel D, Meinl W, Glatt H, Falany CN et al . Potent inhibition of estrogen sulfotransferase by hydroxylated PCB metabolites: a novel pathway explaining the estrogenic activity of PCBs Endocrinology 2000 141: 1897–1900
Safe SH . Polychlorinated biphenyls (PCBs): environmental impact, biochemical and toxic responses, and implications for risk assessment Crit Rev Toxicol 1994 24: 87–149
Falany CN, Xie XW, Wang J, Ferrer J, Falany JL . Molecular cloning and expression of novel sulphotransferase-like cDNAs from human and rat brain Biochem J 2000 346: 857–864
Hume R, Coughtrie MWH, Burchell B . Differential localization of UDP-glucuronosyltransferase in kidney during human embryonic and fetal development Arch Toxicol 1995 69: 242–247
Kakuta Y, Pedersen LG, Carter CW, Negishi M, Pedersen LC . Crystal structure of estrogen sulphotransferase Nature Struct Biol 1997 4: 904–908
Dajani R, Cleasby A, Neu M, Wonacott AJ, Jhoti H, Hood AM et al . X-ray crystal structure of human dopamine sulfotransferase, SULT1A3: molecular modelling and QSAR analysis demonstrate a molecular basis for sulfotransferase substrate specificity J Biol Chem 1999 274: 37862–37868
Bidwell LM, McManus ME, Gaedigk A, Kakuta Y, Negishi M, Pedersen L et al . Crystal structure of human catecholamine sulfotransferase J Mol Biol 1999 293: 521–530
Pedersen LC, Petrotchenko EV, Negishi M . Crystal structure of SULT2A3, human hydroxysteroid sulfotransferase FEBS Lett 2000 475: 61–64
Kakuta Y, Sueyoshi T, Negishi M, Pedersen LC . Crystal structure of the sulfotransferase domain of human heparan sulfate N-deacetylase/N-sulfotransferase 1 J Biol Chem 1999 274: 10673–10676
Pakhomova S, Kobayashi M, Buck J, Newcomer ME . A helical lid converts a sulfotransferase to a dehydratase Nat Struct Biol 2001 8: 447–451
Yoshinari K, Petrotchenko EV, Pedersen LC, Negishi M . Crystal structure-based studies of cytosolic sulfotransferase J Biochem Molec Toxicol 2001 15: 67–75
Kakuta Y, Petrotchenko EV, Pedersen LC, Negishi M . The sulfuryl transfer mechanism: crystal structure of a vanadate complex of estrogen sulfotransferase and mutational analysis J Biol Chem 1998 273: 27325–27330
Dajani R, Hood AM, Coughtrie MWH . A single amino acid, Glu146, governs the substrate specificity of a human dopamine sulfotransferase, SULT1A3 Mol Pharmacol 1998 54: 942–948
Brix LA, Barnett AC, Duggleby RG, Leggett B, McManus ME . Analysis of the substrate specificity of human sulfotransferases SULT1A1 and SULT1A3: site-directed mutagenesis and kinetic studies Biochemistry 1999 38: 10474–10479
Brix LA, Duggleby RG, Gaedigk A, McManus ME . Structural characterization of human aryl sulphotransferases Biochem J 1999 337: 337–343
Petrotchenko EV, Doerflein ME, Kakuta Y, Pedersen LC, Negishi M . Substrate gating confers steroid specificity to estrogen sulfotransferase J Biol Chem 1999 274: 30019–30022
Weinshilboum R . Phenol sulfotransferase inheritance Cell Mol Neurobiol 1988 8: 27–33
Weinshilboum R . Sulfotransferase pharmacogenetics Pharmac Ther 1990 45: 93–107
Weinshilboum R, Aksoy I . Sulfation pharmacogenetics in humans Chem Biol Interact 1994 92: 233–246
Glatt H, Engelke CEH, Pabel U, Teubner W, Jones AL, Coughtrie MWH et al . Sulfotransferases: genetics and role in toxicology Toxicol Lett 2000 112: 341–348
Price RA, Spielman RS, Lucena AL, van Loon JA, Baidak BL, Weinshilboum RM . Genetic polymorphism for human platelet thermostable phenol sulfotransferase (TS PST) activity Genetics 1989 122: 905–914
Reiter C, Weinshilboum R . Platelet phenol sulfotransferase activity: correlation with sulfate conjugation of acetaminophen Clin Pharmacol Ther 1982 32: 612–621
Bonham-Carter SM, Rein G, Glover V, Sandler M, Caldwell J . Human platelet phenol sulphotransferase M and P: substrate specificities and correlation with in vivo sulphoconjugation of paracetamol and salicylamide Br J Clin Pharmacol 1983 15: 323–330
van Loon JA, Weinshilboum RM . Human platelet phenol sulfotransferase: familial variation in thermal stability of the TS form Biochem Genet 1984 22: 997–1014
Jones AL, Roberts RC, Coughtrie MWH . The human phenolsulfotransferase polymorphism is determined by the level of expression of the enzyme protein Biochem J 1993 296: 287–290
Stanley EL, Hume R, Visser TJ, Coughtrie MWH . Differential expression of sulfotransferase enzymes involved in thyroid hormone metabolism during human placental development J Clin Endocrinol Metab 2001 86: 5944–5955
Raftogianis RB, Wood TC, Otterness DM, van Loon JA, Weinshilboum RM . Phenol sulfotransferase pharmacogenetics in humans: association of common SULT1A1 alleles with TS PST phenotype Biochem Biophys Res Commun 1997 239: 298–304
Raftogianis RB, Wood TC, Weinshilboum RM . Human phenol sulfotransferases SULT1A2 and SULT1A1. Genetic polymorphisms, allozyme properties, and human liver genotype–phenotype correlations Biochem Pharmacol 1999 58: 605–616
Nowell S, Ambrosone CB, Ozawa S, MacLeod SL, Mrackova G, Williams S et al . Relationship of phenol sulfotransferase activity (SULT1A1) genotype to sulfotransferase phenotype in platelet cytosol Pharmacogenetics 2000 10: 789–797
Ozawa S, Shimizu M, Katoh T, Miyajima A, Ohno Y, Matsumoto Y et al . Sulfating-activity and stability of cDNA-expressed allozymes of human phenol sulfotransferase, ST1A3*1 (213Arg) and ST1A3*2 (213His), both of which exist in Japanese as well as Caucasians J Biochem 1999 126: 271–277
Li X, Clemens DL, Cole JR, Anderson RJ . Characterization of human liver thermostable phenol sulfotransferase (SULT1A1) allozymes with 3,3′,5′-triiodothyronine as the substrate J Endocrinol 2001 171: 525–532
Raftogianis RB . Human sulfotransferases and cellular response to estrogens and antiestrogens Drug Metab Rev 2001 33: 10
Petrotchenko EV, Pedersen LC, Borchers CH, Tomer KB, Negishi M . The dimerization motif of cytosolic sulfotransferases FEBS Lett 2001 490: 39–43
Kester MHA, Kaptein E, Roest TJ, van Dijk CH, Tibboel D, Meinl W et al . Characterization of human iodothyronine sulfotransferases J Clin Endocrinol Metab 1999 84: 1357–1364
Coughtrie MWH, Gilissen RAHJ, Shek B, Strange RC, Fryer AA, Jones PW et al . Phenol sulphotransferase SULT1A1 polymorphism: molecular diagnosis and allele frequencies in Caucasian and African populations Biochem J 1999 337: 45–49
Frame LT, Gatlin TL, Kadlubar FF, Lang NP . Metabolic differences and their impact on human disease—Sulfotransferase and colorectal cancer Environ Toxicol Pharmacol 1997 4: 277–281
Bamber DE, Fryer AA, Strange RC, Elder JB, Deakin M, Rajagopal R et al . Phenol sulphotransferase SULT1A1*1 genotype is associated with reduced risk of colorectal cancer Pharmacogenetics 2001 11: 679–685
Seth P, Lunetta KL, Bell DW, Gray H, Nasser SM, Rhei E et al . Phenol sulfotransferases: hormonal regulation, polymorphism, and age of onset of breast cancer Cancer Res 2000 60: 6859–6863
Zheng W, Xie DW, Cerhan JR, Sellers TA, Wen WQ, Folsom AR . Sulfotransferase 1A1 polymorphism, endogenous estrogen exposure, well-done meat intake, and breast cancer risk Cancer Epidem Biomar 2001 10: 89–94
Chou HC, Lang NP, Kadlubar FF . Metabolic activation of the N-hydroxy derivative of the carcinogen 4-aminobiphenyl by human tissue sulfotransferases Carcinogenesis 1995 16: 413–417
Steiner M, Bastian M, Schulz WA, Pulte T, Franke KH, Rohring A et al . Phenol sulphotransferase SULT1A1 polymorphism in prostate cancer: lack of association Arch Toxicol 2000 74: 222–225
Kalow W, Bertilsson L . Interethnic factors affecting drug response Advanc Drug Res 1994 23: 1–53
Nebert DW, Menon AG . Pharmacogenomics, ethnicity, and susceptibility genes Pharmacogenomics J 2001 1: 19–22
Zhu X, Veronese ME, Iocco P, McManus ME . cDNA cloning and expression of a new form of human aryl sulfotransferase Int J Biochem Cell Biol 1996 28: 565–571
Engelke CEH, Meinl W, Boeing H, Glatt H . Association between functional genetic polymorphisms of human sulfotransferases 1A1 and 1A2 Pharmacogenetics 2000 10: 163–169
Dooley TP, Haldeman-Cahill R, Joiner J, Wilborn TW . Expression profiling of human sulfotransferase and sulfatase gene superfamilies in epithelial tissues and cultured cells Biochem Biophys Res Commun 2000 277: 236–245
Carlini EJ, Raftogianis RB, Wood TC, Jin F, Zheng W, Rebbeck TR et al . Sulfation pharmacogenetics: SULT1A1 and SULT1A2 allele frequencies in Caucasian, Chinese and African–American subjects Pharmacogenetics 2001 11: 57–68
Iida A, Sekine A, Saito S, Kitamura Y, Kitamoto T, Osawa S et al . Catalog of 320 single nucleotide polymorphisms (SNPs) in 20 quinone oxidoreductase and sulfotransferase genes J Hum Genet 2001 46: 225–240
Freimuth RR, Eckloff B, Wieben ED, Weinshilboum RM . Human sulfotransferase SULT1C1 pharmacogenetics: gene resequencing and functional genomic studies Pharmacogenetics 2001 11: 747–756
Freimuth RR, Raftogianis RB, Wood TC, Moon E, Kim UJ, Xu J et al . Human sulfotransferases SULT1C1 and SULT1C2: cDNA characterization, gene cloning, and chromosomal localization Genomics 2000 65: 157–165
Yoshinari K, Nagata K, Shimada M, Yamazoe Y . Molecular characterization of ST1C1-related human sulfotransferase Carcinogenesis 1998 19: 951–953
Li XY, Clemens DL, Anderson RJ . Sulfation of iodothyronines by human sulfotransferase 1C1 (SULT1C1) Biochem Pharmacol 2000 60: 1713–1716
Song W-C, Qian YM, Li AP . Estrogen sulfotransferase expression in the human liver: marked interindividual variation and lack of gender specificity J Pharmacol Exp Ther 1998 284: 1197–1202
Rotter JI, Wong FL, Lifrak ET, Parker LN . A genetic component to the variation of dehydroepiandrosterone sulfate Metabolism 1985 34: 731–736
Aksoy IA, Sochorová V, Weinshilboum RM . Human liver dehydroepiandrosterone sulfotransferase—nature and extent of individual variation Clin Pharmacol Ther 1993 54: 498–506
Wood TC, Her C, Aksoy I, Otterness DM, Weinshilboum RM . Human dehydroepiandrosterone sulfotransferase pharmacogenetics: quantitative western analysis and gene sequence polymorphisms J Steroid Biochem Mol Biol 1996 59: 467–478
Nagata K, Yamazoe Y . Pharmacogenetics of sulfotransferase Annu Rev Pharmacol Toxicol 2000 40: 159–176
Thomae BA, Eckloff BW, Freimuth RR, Wieben ED, Weinshilboum RM . Human sulfotransferase SULT2A1 pharmacogenetics: genotype-to-phenotype studies Pharmacogenomics J 2002 2: 48–56
Ingelman-Sundberg M . Implications of polymorphic cytochrome P450-dependent drug metabolism for drug development Drug Metab Dispos 2001 29: 570–573
Hodgson J . ADMET—turning chemicals into drugs Nat Biotechnol 2001 19: 722–726
Funk C, Ponelle C, Scheuermann G, Pantze M . Cholestatic potential of troglitazone as a possible factor contributing to troglitazone-induced hepatotoxicity: In vivo and in vitro interaction at the canalicular bile salt export pump (Bsep) in the rat Mol Pharmacol 2001 59: 627–635
Keppler D, Kamisako T, Leier I, Cui Y, Nies AT, Tsujii H et al . Localization, substrate specificity, and drug resistance conferred by conjugate export pumps of the MRP family Adv Enzyme Regul 2000 40: 339–349
Suzuki H, Sugiyama Y . Transport of drugs across the hepatic sinusoidal membrane: sinusoidal drug influx and efflux in the liver Semin Liver Dis 2000 20: 251–263
Kullak-Ublick GA, Stieger B, Hagenbuch B, Meier PJ . Hepatic transport of bile salts Semin Liver Dis 2000 20: 273–292
Qian YM, Sun XJ, Tong MH, Li XP, Richa J, Song WC . Targeted disruption of the mouse estrogen sulfotransferase gene reveals a role of estrogen metabolism in intracrine and paracrine estrogen regulation Endocrinology 2001 142: 5342–5350
Song W-C, Qian Y, Sun X, Negishi M . Cellular localization and regulation of expression of testicular estrogen sulfotransferase Endocrinology 1997 138: 5006–5012
Borthwick EB, Burchell A, Coughtrie MWH . Differential expression of hepatic estrogen, phenol and dehydroepiandrosterone sulfotransferases in genetically-obese diabetic (ob/ob) male and female mice J Endocrinol 1995 144: 31–37
Meloche CA, Falany CN . Expression of SULT2B1b in hormonally regulated human tissues Drug Metab Rev 2001 33: 230
Geese WJ, Raftogianis RB . Biochemical characterization and tissue distribution of human SULT2B1 Biochem Biophys Res Commun 2001 288: 280–289
Javitt NB, Lee YC, Shimizu C, Fuda H, Strott CA . Cholesterol and hydroxycholesterol sulfotransferases: identification, distinction from dehydroepiandrosterone sulfotransferase, and differential tissue expression Endocrinology 2001 142: 2978–2984
Ozawa S, Tang Y-M, Yamazoe Y, Kato R, Lang NP, Kadlubar FF . Genetic polymorphism in human liver phenol sulfotransferases involved in the bioactivation of N-hydroxy derivatives of carcinogenic arylamines and heterocyclic amines Chem Biol Interact 1998 109: 237–248
Acknowledgements
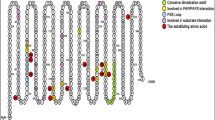
I am grateful to all the members of my laboratory (past and present), and to numerous valued collaborators, who have contributed to the work and ideas described here. I am particularly indebted to Dr Rebecca Raftogianis (Fox Chase Cancer Center, Philadelphia, USA) for her critical reading of the manuscript and for helpful suggestions regarding nomenclature, and to Professor Jyrki Taskinen (University of Helsinki, Finland) for producing the model upon which Figure 2 is based. Current research in the laboratory is supported by the Medical Research Council, the Commission of the European Communities (QLG3-CT-2000-00930), Tenovus Scotland, the Scottish Office Chief Scientist Office and the Human Drug Conjugation Consortium.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coughtrie, M. Sulfation through the looking glass—recent advances in sulfotransferase research for the curious. Pharmacogenomics J 2, 297–308 (2002). https://doi.org/10.1038/sj.tpj.6500117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.tpj.6500117
Keywords
This article is cited by
-
Association of CHEK2 I157T and SULT1A1 R213H genetic variants with risk of sporadic colorectal cancer in a sample of Egyptian patients
Egyptian Journal of Medical Human Genetics (2022)
-
A simple method to measure sulfonation in man using paracetamol as probe drug
Scientific Reports (2021)
-
Bio-artificial bone formation model with a radial-flow bioreactor for implant therapy—comparison between two cell culture carriers: porous hydroxyapatite and β-tricalcium phosphate beads
Human Cell (2019)
-
Are some people at increased risk of paracetamol-induced liver injury? A critical review of the literature
European Journal of Clinical Pharmacology (2018)
-
On the Molecular Basis Underlying the Metabolism of Tapentadol Through Sulfation
European Journal of Drug Metabolism and Pharmacokinetics (2017)